Clinical Development of Odetiglucan
Odetiglucan, in combination with multiple therapies, has demonstrated encouraging clinical activity across diverse tumor histologies, including in patients with liver metastases. Odetiglucan is currently being evaluated in two Phase 2 investigator-initiated clinical trials in combination with an immune checkpoint inhibitor and another myeloid agonist in patients with colorectal and pancreatic cancers.
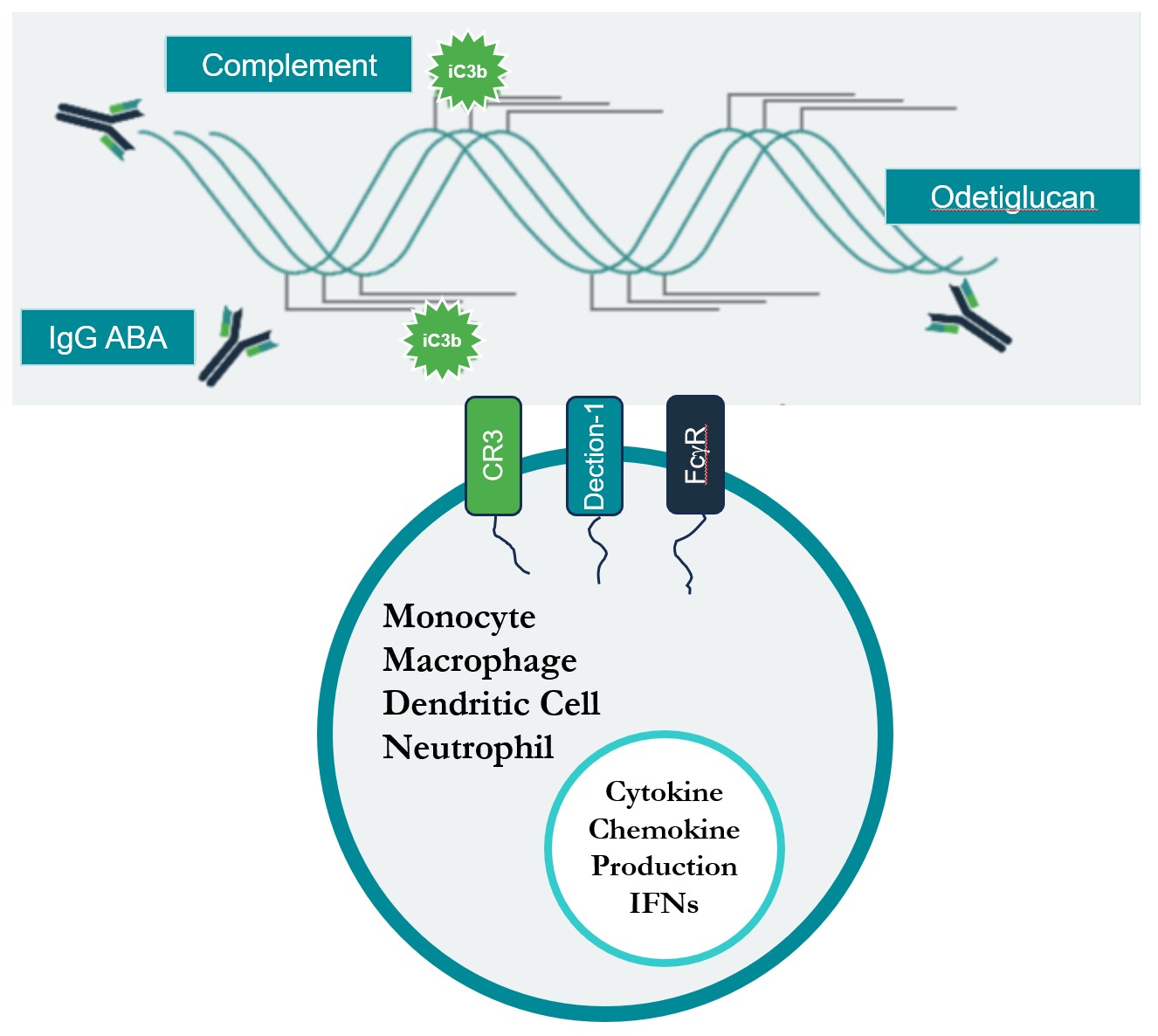
The Odetiglucan Immune Complex
Odetiglucan is an immune agonist that modulates both the innate and adaptive immune systems. It forms an immune complex in vivo through three receptors - Dectin-1, FcɣR, and CR3 - resulting in the activation of anti-tumor immunity.
Unlike many other PRR agonists (cGAS-STING, TLR, etc.), Odetiglucan has been clinically observed to be generally well-tolerated and suitable for systemic administration due to its non-inflammatory, chemokine-driven innate immune activation. Consequently, Odetiglucan is not restricted to intratumoral injection and does not rely on an abscopal effect to achieve systemic immune modulation.
Odetiglucan drives anti-tumor immunity by reprogramming the immunosuppressive tumor microenvironment, enhancing antigen presentation, priming a de-novo T cell responses, and inducing trained immunity to promote durable immune memory.